Introduction
The implantation rate of devices such as pacemakers (PMs) and implantable cardioverter–defibrillators (ICDs), with or without cardiac resynchronization therapy (CRT) options, has significantly increased over the last few decades,1–3 leading to a rising number of failed leads, device-related infections, and lead or device recalls. Consequently, the need for PM and/or ICD lead extractions is increasing.4
Recently introduced PM leads can typically be removed by direct traction. However, chronically implanted leads become encapsulated by fibrotic attachments.5–8 Adhesions not only occur at the lead tip but also are commonly found along any length of lead where a contact between the lead and vein, valve, or endocardial structures such as the superior vena cava exists.5–10 Over the last 20 years, specific tools and techniques for transvenous lead extraction have been developed to assist in freeing the lead body from the adhesions as well as the lead tip from the myocardium, to prevent laceration of the myocardium and to provide enough room for the lead to be withdrawn while preventing its disintegration. Specialized tools include locking stylets, telescoping sheaths with or without additional cutting capability (e.g., metal composition, excimer laser, and radiofrequency current), snares, grasping devices, or other devices used to engage or entrap and remove lead fragments. The current review aims to provide an overview of lead extraction, including the success and complication rates of different extraction methods currently available.
Definition of lead and system extraction
Within the general category of “lead removal,” distinctions must be made between simple procedures that can be performed via the implant vein without specialized tools (“lead explant”) and removal of leads involving more complex procedures (“lead extraction”).11 The definitions are as follows:
Lead removal: Removal of a pacing or defibrillator lead using any technique
Lead explant: Lead removal using simple traction techniques (no locking stylet, telescoping sheaths, or femoral extraction tools)
Lead extraction: Removal of a lead that has been implanted for more than 1 year, removal of a lead, regardless of the duration of implant, that requires the assistance of specialized equipment that is not included as part of the typical implant package, and/or removal of a lead through a route other than via the implant vein
ICD leads may require specialized extraction equipment even when the duration of implantation is less than 1 year.
Indications for lead extraction
The most common indication for extraction is PM infection, including pocket infection or endocarditic vegetation involving any intravascular component of the PM system. However, other important common indications include lead malfunction (fracture or failure), lead or device erosion, lead upgrade, retained lead or lead fragment causing potentially life-threatening arrhythmias, thromboembolic complications or venous obstruction, device recall, and a lead that interferes with the operation of another implanted device (e.g., PM or ICD). Recently published guidelines can be found in the Heart Rhythm Society Expert Consensus on Lead extraction (Table 1).11
Table 1.
Indications for lead extraction
Tools, technique and outcomes for lead removal
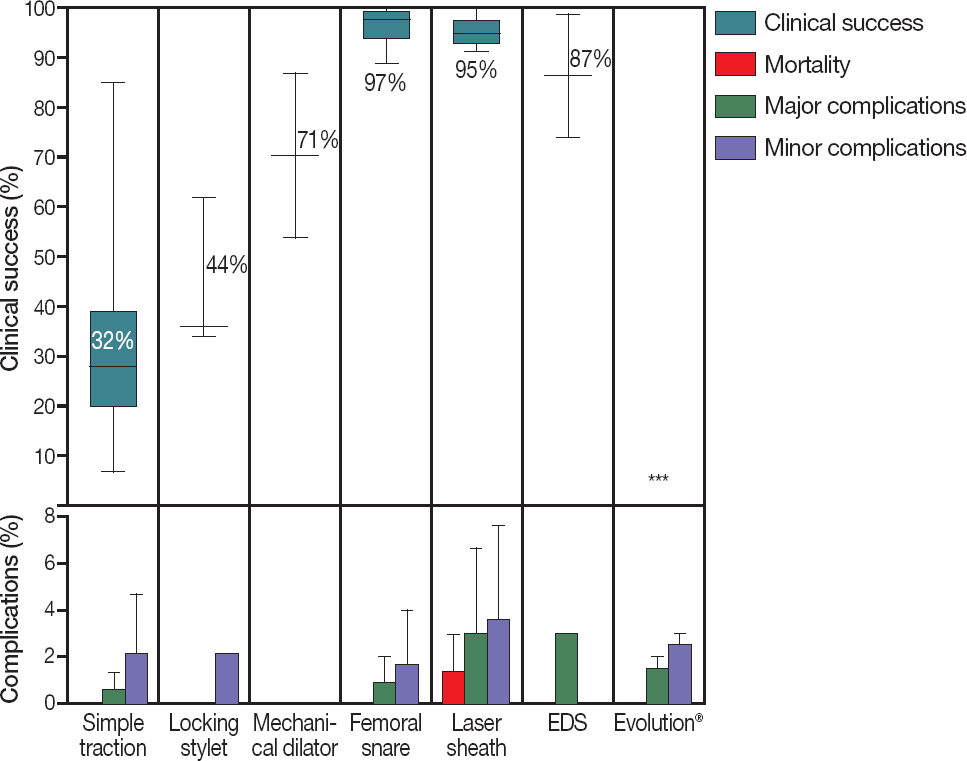
Most lead extractions are achieved through a percutaneous transvenous approach. However, the following are the three main indications for open surgical extraction: failed percutaneous lead extraction, surgical unrelated cardiac disease (such as pre-existing need for valve replacement), and endocarditis with very large infected vegetations that would require open extraction to minimize embolic complications. The clinical outcomes of different extraction methods are presented in Figure 1.12
Figure 1.
Clinical outcomes of different extraction methods. Clinical success rate is reported per lead and complication rate is reported per patient. The percentages represent the mean success rate. *Clinical success or complication rates were not reported for this extraction method. EDS, Electrosurgical dissection sheath
Simple traction
The first attempt to extract a transvenous PM or ICD lead usually involves simple traction. Simple traction has been performed for almost 40 years and is the most basic technique for lead removal. It can be combined with graded traction and/or rotational forces. This technique of applying traction to the lead in combination with the use of tools typically supplied for lead implantation (non-locking stylets, fixation screw retraction clips) is particularly successful in leads with recently implanted leads.13 The success rate of TLE by simple traction was observed in 9–31% (median, 19%) of patients and in 7–85% (median, 28%) of leads. The success rates of simple traction vary considerably among different studies.12 No simple traction-related deaths were reported in the 3,769 patients in whom it was performed.
The main limitation of manual traction is its limited success in completely removing older leads. The forceful traction may sever the insulation and conductor coils of the leads, resulting in denuded retained lead fragments. Major complications, including three cases of cardiac avulsion requiring surgical intervention, occurred in 0–1.3% of patients (Figure 1).12
Non-powered traction tools
Over time, leads become adherent to either the myocardium or vascular walls in varying degrees owing to encapsulating fibrotic tissue at the contact sites. Thus, during application of traction to chronically implanted leads, force is distributed over all fibrotic binding sites and weakened at the distal end of the lead. Non-powered tools are developed to direct the force of traction to the length or at the distal end of the lead (locking stylets) or to disrupt and dilate the encapsulating fibrotic tissue (mechanical dilator sheaths).
Locking stylets
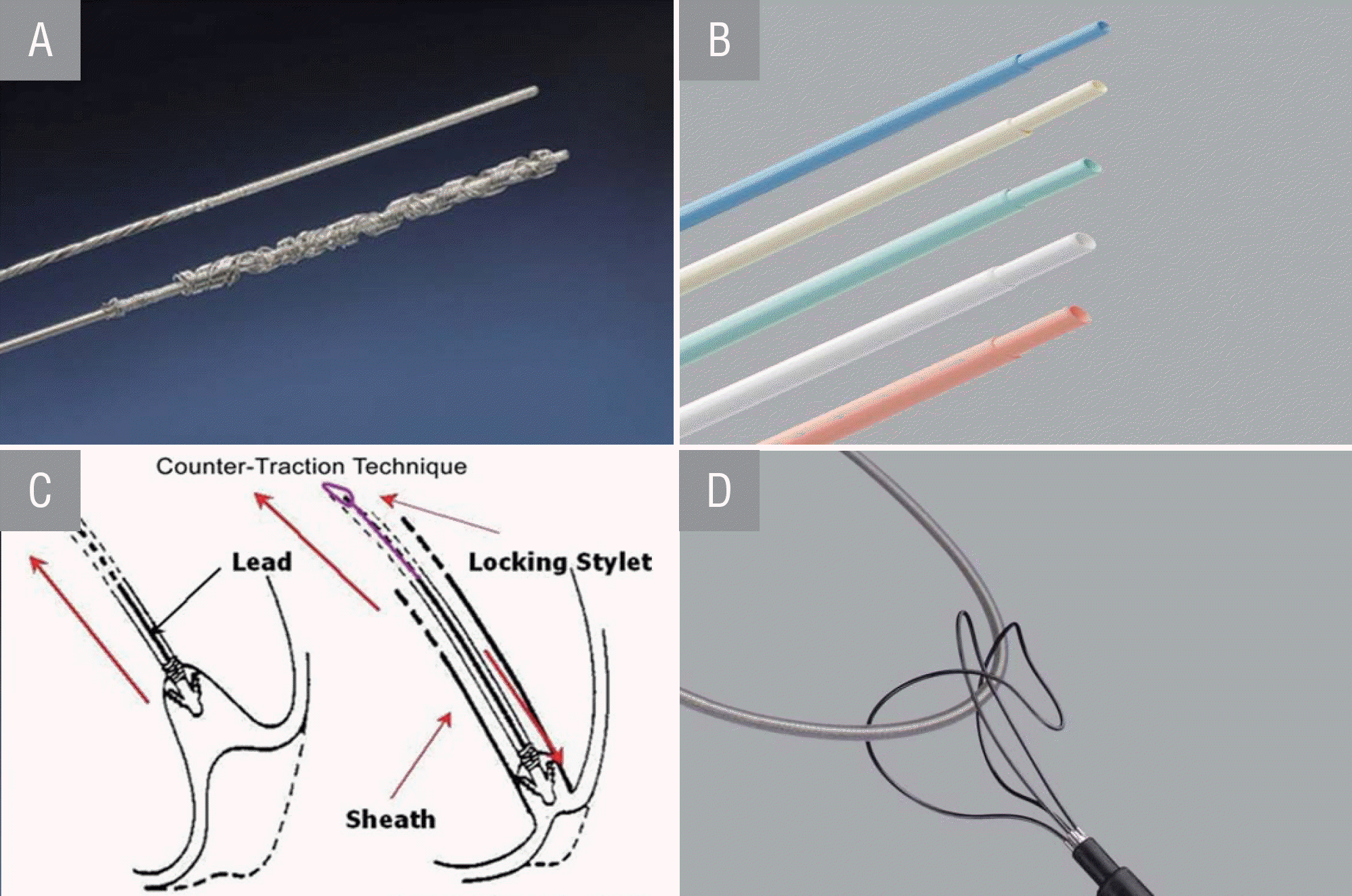
This technique uses a special traction device to minimize lead disintegration. A locking stylet is introduced into the central lumen of the lead where it can be locked into the coil close to the lead tip or anywhere along the conductor coil (Figure 2A). The improved traction helps prevent elongation of the lead body and coil during exertion. The TLE success rate after the use of both simple traction and locking stylets was observed in 22–85% (median, 43%) of patients and in 34–62% (median, 36%) of leads.12 No procedure-related deaths due to TLE using locking stylets were reported (Figure 1).
Figure 2.
A, Liberator® locking stylet. B, Byrd dilator sheaths telescoping polypropylene. C, Counter-traction technique. D, Needle's eye snare.
Important limitations to the use of a locking stylet exist. A broken conductor or distorted central lumen renders it impossible to introduce the stylet. Excessive force can dislocate the stylet, or the distal conductor coil can still unwind or even disconnect from the electrode.
Mechanical dilator sheaths
A common third step in the stepwise approach of lead extraction is the use of a mechanical dilator sheath. A telescoping sheath, available in different materials (i.e., polypropylene, Teflon®, or stainless steel), is advanced along the lead to disrupt and dilate the encapsulating fibrotic attachments. A locking stylet is still required to enable countertraction as the sheath is advanced. On reaching the distal electrode, the larger bore outer sheath is positioned and held against the myocardium to prevent its inversion during traction on the locking stylet. The force is, therefore, applied at the adherent scar without gross displacement of the myocardium (Figure 2B).
Cecchin et al.14 reported that after a mean lead dwell time of 7 years, 20% of the leads were successfully extracted using dilator sheaths, resulting in an overall success of 54% after simple traction, locking stylet, and mechanical sheath use. Results using conventional sheaths also include the use of a femoral approach, and are reported in the U.S. Lead Extraction Database.15 Of the 2,195 leads, 86.8% were completely removed, whereas 7.5% were partially removed. Although countertraction prevents invagination of the myocardium, perforation of the myocardium is still possible. The lead tip may become incorporated into the myocardium, leading to perforation after dislocation of the tip. Moreover, the possibility of increasing the force using countertraction can lacerate the myocardium, especially in the thin-walled atrium.16
Femoral and transjugular extraction techniques
A transfemoral approach is helpful when access or extraction via the implanting vein is impossible or when the leads are fractured or cut. The Needle's Eye Snare is the most frequently used tool for lead extractions via the transfemoral approach. Usually, the use of femoral snare is the last step in TLE (Figure 2D). Bracke et al.17 reported the results of the transfemoral approach in a large cohort of patients, and the mean dwell time of the leads was 7.6 years. After simple traction had failed, 98% of the remaining leads were successfully extracted using the Needle's Eye Snare. No procedure-related deaths were observed, major complications were observed in 0.6% of the patients (two patients with cardiac avulsion requiring surgical intervention), and no minor complications occurred.
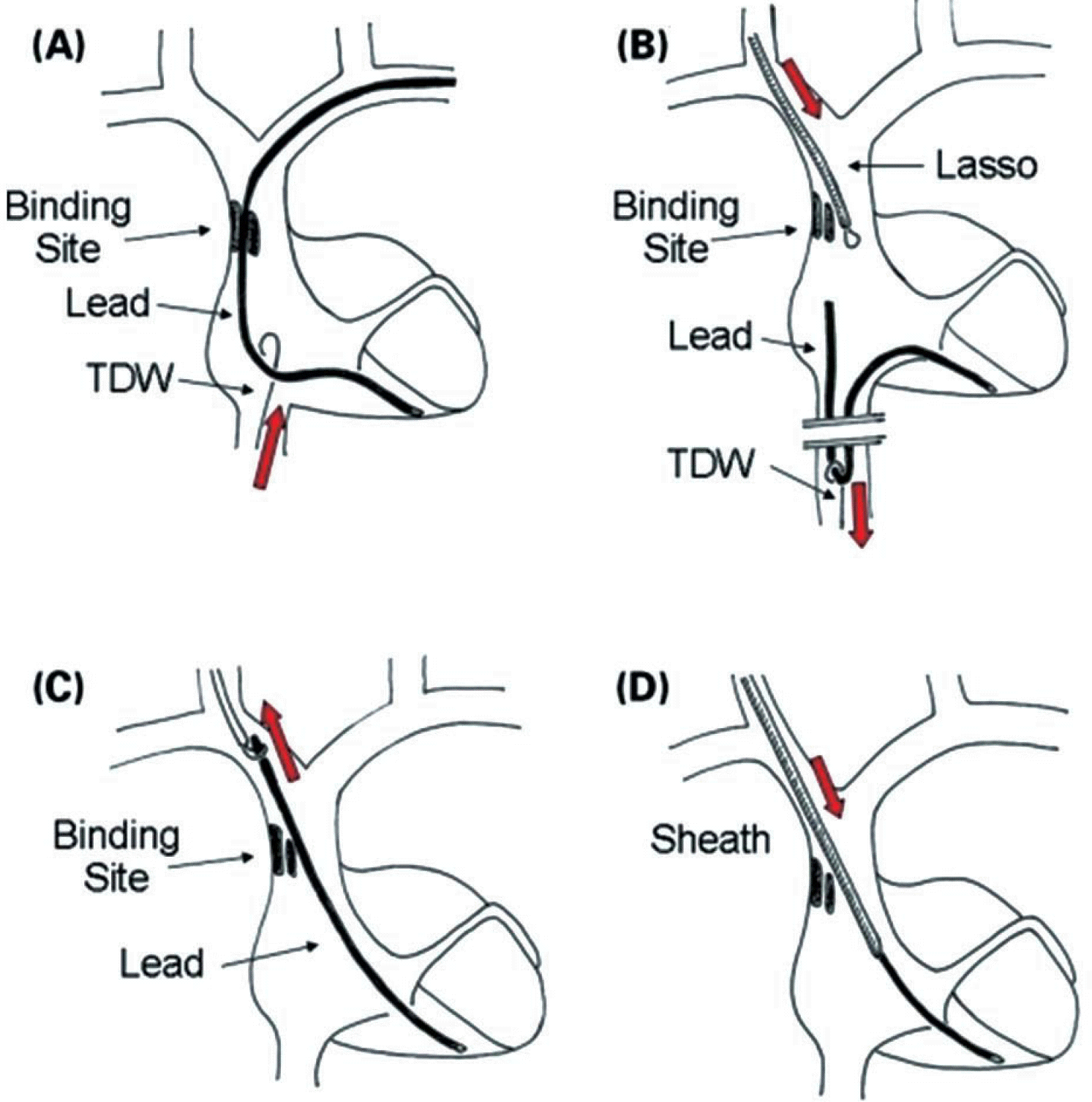
The transjugular approach enables the removal of previously cut leads that have retracted into the central venous circulation and/or have been damaged during an extraction performed via a superior approach. In a large single-center study by Bongiorni et al.,18 the TLE approach using simple traction, dilator sheaths, and/or a femoral snare was successful in 89% of the leads. However, extraction of the remaining leads was attempted via a transjugular approach, resulting in a total success rate of 99%. Consecutive steps of the internal transjugular approach are presented in Figure 3.18
Figure 3.
Consecutive steps of the internal transjugular approach (ITA) in case of crossover from the venous entry approach (VEA). (A) A tip deflecting wire is advanced via the femoral vein in order to assess the possibility to grasp the lead and to move it. (B) Once the lead has been grasped, it is pulled down in the inferior vena cava and slipped through the binding site; a Lasso, introduced through the internal jugular vein, is advanced near the proximal end of the lead. (C) The lead is caught by the Lasso, pulled up and exposed through the jugular vein. (D) Dilatation using a dilating sheath is performed. See the text for further details. TDW, tip deflecting wire.
Powered traction tools
Non-po wered sheaths use blunt dissection, while powered extraction sheaths use an energy source to disrupt adhesions between the lead and the endothelial or endocardial wall. Powered sheaths include laser sheaths, electrosurgical dissection sheaths, and rotating threaded tip sheaths. Powered traction tools are usually applied during simple traction, while non-powered traction tools fail in a stepwise approach.
Laser sheaths
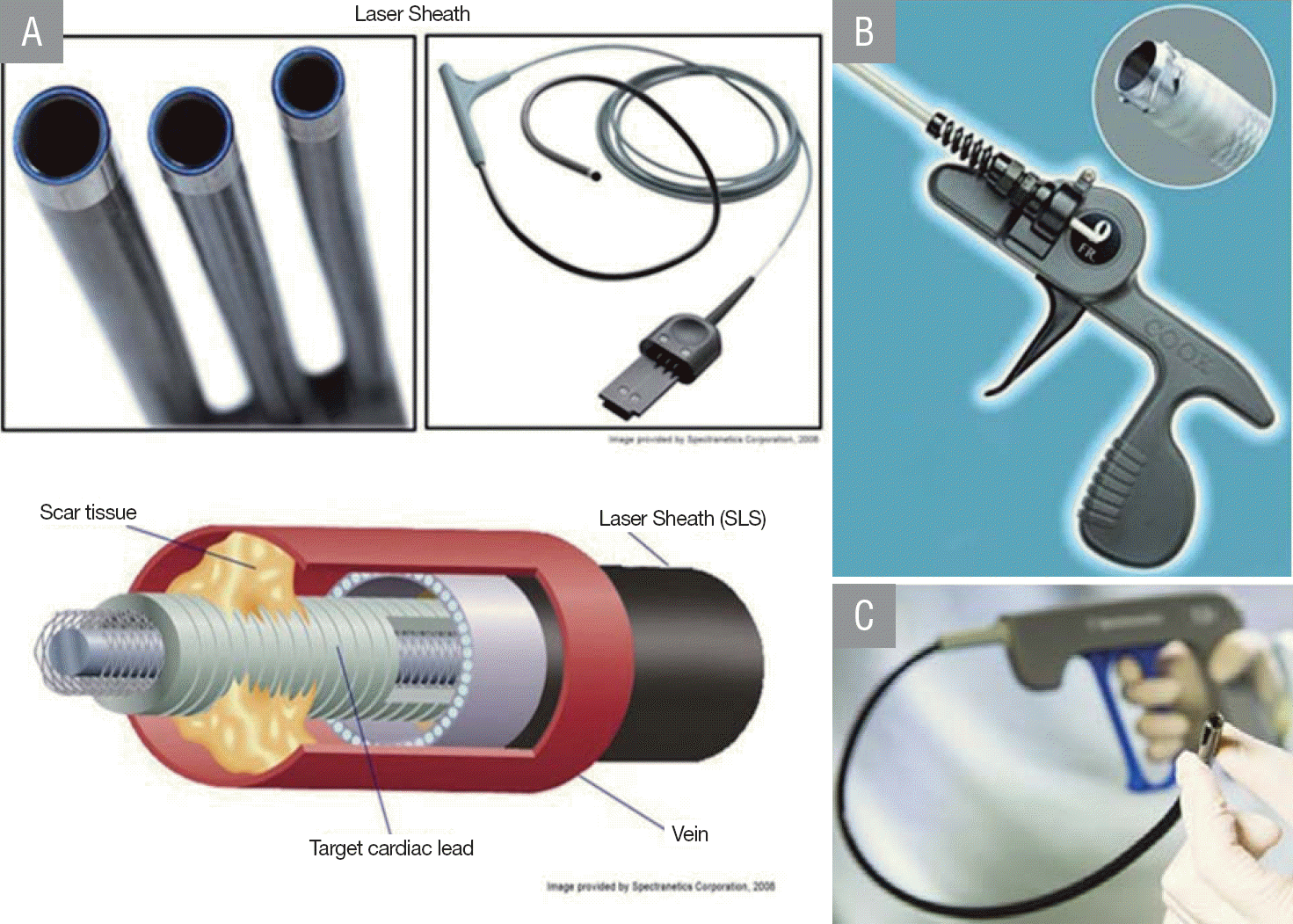
A laser sheath is a tubular structure that passes over the PM lead. It consists of a thin layer of optical fibers sandwiched between polymer walls. The distal tip delivers a ring of laser light in pulses to a tissue depth of 100 μm, so that only the tissue immediately in contact with the sheath tip dissolves. Thus, the fibrous tissue encapsulating the lead body is removed in a controlled manner, and occluded vasculature can be re-canalized (Figure 4A). Studies in which patients underwent laser sheath extraction demonstrated clinical success after stepwise approach in 85–96% (median, 94%) of patients and 93–100% (mean, 95%) of leads (lead age range, 1.1–6.0 years). Procedure-related mortality was observed in 0–2.7% of the patients, with major and minor complications in 0–7.3% and 0–8.0% of the patients, respectively (Figure 1).12
Electrosurgical dissection sheath
The electrosurgical dissection sheath utilizes radiofrequency energy, similar to the cautery tool used in surgery, to cut through fibrous tissue. Two electrodes are exposed at the tip of the sheath, which allows linear dissection of adhesions comparable to a cautery tool. As opposed to the laser sheath, the electrosurgical dissection sheath permits localized application of energy instead of circumferential dissection.
Rotating threaded tip sheaths
The most recent addition to the equipment for the lead extractionist is a “hand-powered” rotating threaded tip sheath. This sheath is attached to a handle that controls the rotation of a threaded screw mechanism at the tip of the sheath, causing it to bore through adhesions around the lead. The Evolution® and Spectranetics TightRail® rotating dilator sheaths are currently the only rotating threaded tip sheaths available, and they have been advocated as especially advantageous in disrupting calcified fibrosis (Figure 4B and 4C).19 The Evolution tool showed an overall success rate of 88%, with an average lead age of 7.1 years. Major complications occurred in 0.7–1.5% of the patients, with vascular tear requiring surgery.20,21
ICD leads
The shock coils of high-voltage ICD leads allow fibrous ingrowth, resulting in dense vascular and myocardial adhesions.23 Therefore, compared with regular PM leads, ICD leads might pose an additional challenge and could have an increased risk of complications. Chronically implanted dual-coil ICD leads can pose the greatest challenge to extraction because the proximal externalized coil attracts additional adhesions at the level of the superior vena cava, which further augments the risk of venous tear during extraction. This makes the use of single-coil leads at implant an attractive option, particularly in younger patients who are more likely to require revisions during their lifetime. In experienced hands, ICD lead extractions have been reported to have an overall success rate of 88–100% (median, 99%) of leads or 98–100% (median, 96%) of patients.
Coronary sinus lead extraction
Limited experience regarding CS lead extraction has been reported, but initial reports24,25 suggest that the vast majority of these leads can be safely removed. Simple traction of the lead is often successful; however, concerns exist regarding the perforation of the tortuous and fragile CS and complications in the often frail CRT recipients.
Complications of lead extraction
Most complications can be defined and/or classified according to the level of their severity (Table 2). Complication rates among various intravascular extraction techniques are comparable. All techniques (except direct traction) rely on countertraction; hence, tamponade resulting from perforation after dislocation of the lead tip is common to all these techniques. Analysis of data from a registry of 5,339 lead extractions over 10 years revealed a major complication rate of 1.6%.16 The predictors of risk for complications include the experience of the treating physician, requirement of a large number of leads for extraction, long implant duration of the oldest lead, ICD lead removal, and use of laser.
Table 2.
Definition and classification of complications
Facilities, training and equipment required for safe lead extraction Lead extraction procedures should ideally be performed in hospitals with onsite facilities for cardiothoracic surgery. The surgical team must be readily available with the necessary equipment and facilities for rapid thoracotomy and sternotomy.
Given the technical challenges and risk of life-threatening complications, clinicians who wish to perform this procedure should be adequately trained in extraction techniques and management of complications. They should seek training only in hospitals that have sufficient procedural volume of device implantations and extractions to maintain the skills of the physician and the team.
Conclusion
Transvenous lead extraction includes invasive techniques primarily designed to address concerns related to cardiac pacemaker and ICD lead management. The number of lead extractions continues to rise, and is likely to increase further in the foreseeable future, with the ever-increasing CRM implants and extraction indications. No single technique is sufficient to address all extractions, and centers performing extractions should have the necessary expertise and equipment to perform different techniques depending on the clinical scenario. A stepwise extraction approach can result in a clinically successful TLE in up to 100% of the leads, with a relatively low risk of procedure-related mortality and complications. In case of a lack of a local physician and institutional expertise, the patient should be referred to a center with appropriate facilities. The future of lead extraction would benefit from recommendations regarding extraction approaches and the timing of crossover to a different extraction method to further improve success rates and prevent unnecessary complications.